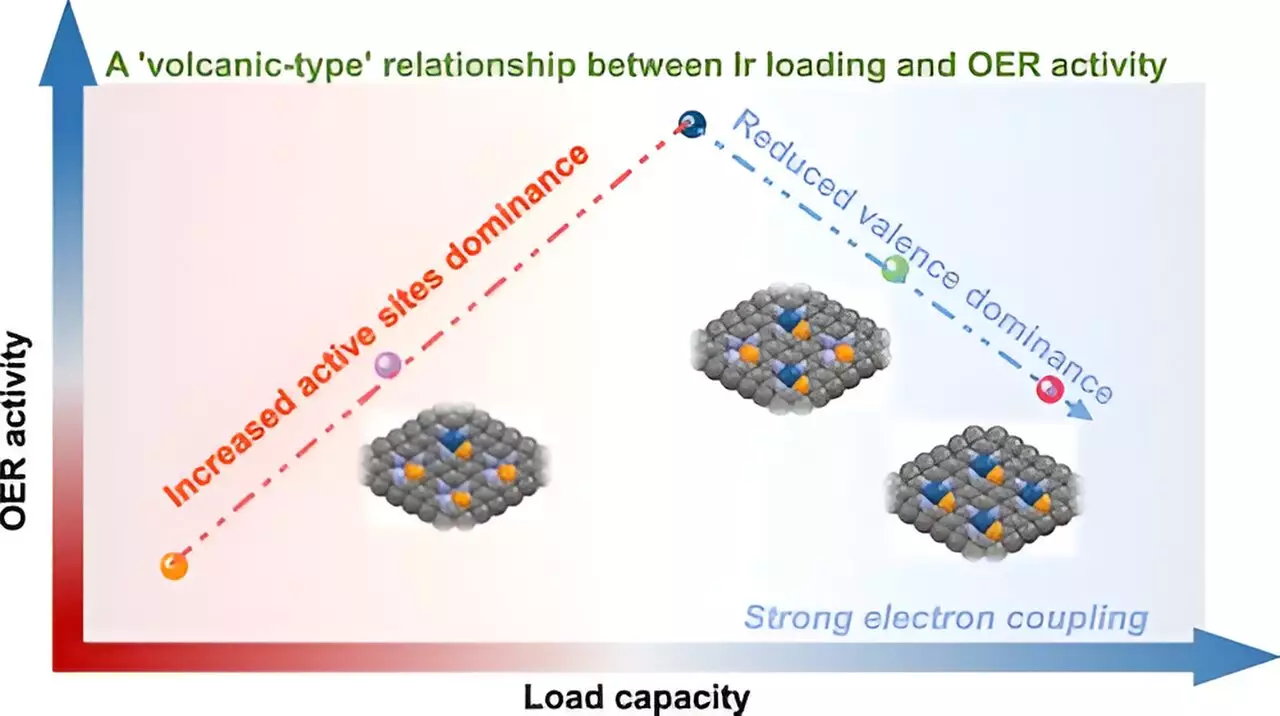
Recent advances in the realm of catalysis have placed single-atom catalysts (SACs) at the forefront of scientific innovation, particularly due to their exceptional efficiency and customizable properties. A study led by Prof. Yan Wensheng at the University of Science and Technology of China (USTC) sheds light on a phenomenon termed the “volcano-type” relationship between metal loading and the activity of acidic oxygen evolution reactions (OER) in SACs. The findings promise a significant leap forward, addressing long-standing challenges in achieving optimal performance in these catalysts.
The Challenge of Metal Loading
One of the central hurdles in single-atom catalysis is the fabrication of metal catalysts that can sustain high metal loadings while maintaining atomic-level dispersion. This balance is crucial, as excessive clustering of metal atoms can undermine the catalytic potential of the material. Prof. Wensheng’s team adopted a novel P-anchoring strategy, culminating in the synthesis of iridium-based SACs (x-IrPN3@C) with loadings spanning from 5% to an impressive 21wt%. This innovative approach not only demonstrated the potential for higher metal loadings but also highlighted the importance of structural coordination in stabilizing metal atoms to prevent aggregation.
Clarifying the Volcano-Type Relationship
Through sophisticated techniques such as synchrotron radiation XAS spectroscopy and X-ray photoelectron spectroscopy (XPS), the researchers unraveled the complex dynamics at play in the “volcano-type” relationship. Unlike a straightforward correlation where increased metal loading typically results in enhanced activity, the research unveiled a more nuanced behavior: initially, as iridium loading increases, the number of available catalytic sites surges, boosting the efficiency of OER. However, once the iridium concentration hits a critical threshold, the interaction between neighboring atoms leads to a detrimental effect—diminishing the valence state of iridium and ultimately reducing catalytic efficacy.
The Implications for Future Catalytic Design
This groundbreaking understanding of the intricate interactions at high metal loadings has far-reaching implications for the design of future catalysts. Instead of merely ramping up metal loadings in a bid for enhanced activity, researchers can now take a more strategic approach, optimizing metal arrangements to harness the benefits of SACs while avoiding potential pitfalls. This insight aligns with broader trends in catalysis research, which increasingly emphasizes precision engineering over brute-force methods.
In essence, the study encapsulates a pivotal moment in the field of catalysis, stripping away the simplistic paradigms that have traditionally guided researchers. The revelations regarding the interactions of metal atoms not only deepen our understanding of single-atom catalysts but also set the stage for the development of more efficient and economically viable catalytic solutions in various applications—from renewable energy production to advanced materials synthesis. This could signify a transformative leap towards unlocking the full potential of SACs in addressing some of the world’s pressing energy challenges.