In modern industrial society, the need for efficient pollutant reduction methods has never been more urgent. Traditional approaches to mitigating hydrocarbon emissions often involve high energy input, which can exacerbate environmental issues rather than alleviate them. The reliance on outdated catalysts has posed significant challenges, leading to calls for new breakthroughs in the field of organic chemistry. This urgent demand has fueled a recent innovative endeavor in catalyst development that promises substantial advancements in our fight against harmful emissions.
An Exciting Advancement at UNIST
Recently, a team of researchers at the Ulsan National Institute of Science and Technology (UNIST) made significant strides in environmental chemistry by engineering a novel catalyst capable of closely mimicking the oxidation processes of natural metalloenzymes. Under the leadership of Professor Jaeheung Cho, this innovative research has managed to define a new frontier for catalysts utilized in breaking down harmful hydrocarbons. Published in the prestigious Journal of the American Chemical Society, this groundbreaking work emphasizes the potential for achieving superior results with fewer resources and reduced energy expenditures.
How It Works: Mimicking Nature
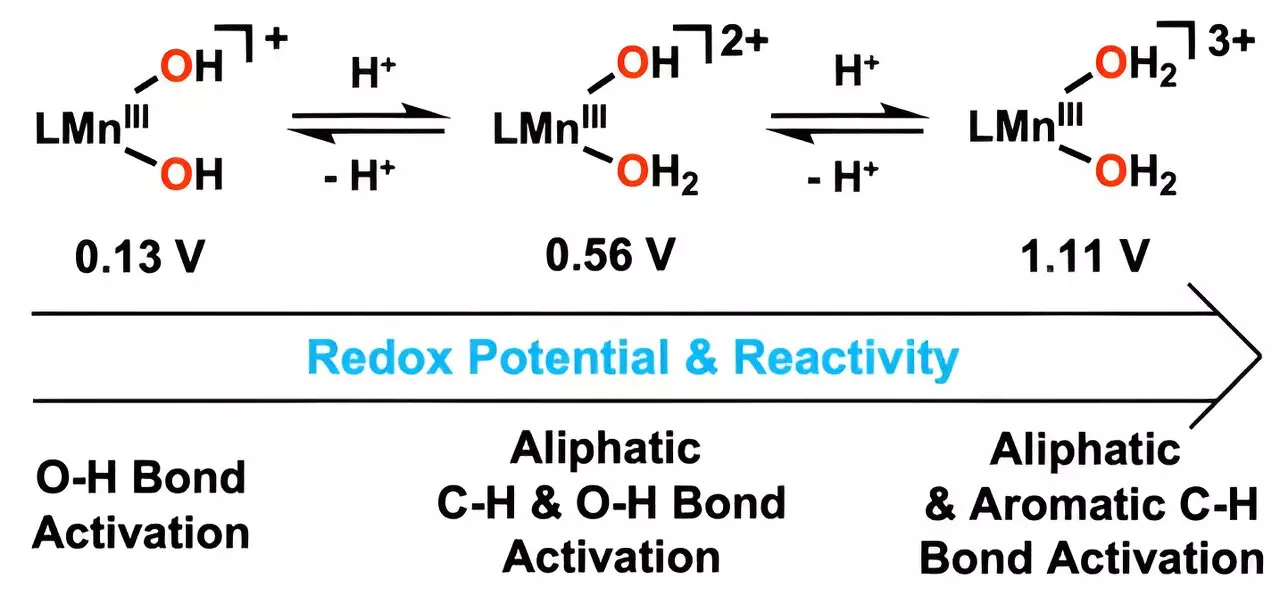
The ingenuity of this new catalyst lies in its composition and design, particularly in manipulating the oxidation process. By integrating hydrogen ions into the hydroxo ligand and employing metal-bound water molecules, the researchers have developed a system that effectively oxidizes carbon-hydrogen (C–H) bonds at surprisingly low temperatures. This strategy is not merely an incremental change; it represents a significant paradigm shift for chemical processes involved in hydrocarbon degradation. The scientists have successfully harnessed nature-inspired mechanisms to create a catalyst that is not only powerful but also efficient and sustainable.
Performance and Applications
The real-world implications of this research are noteworthy. The new catalyst excels at oxidizing anthracene, a compound notorious for its stubborn carbon-hydrogen bonds. By achieving this at low temperatures, the team underscores the potential for safer and more energy-efficient industrial applications, broadening horizons for tackling a wide array of toxic compounds. Furthermore, its capacity to decompose aromatic hydrocarbons—often extremely stable and insoluble in water—highlights how this catalyst could pave the way toward resolving issues posed by persistent environmental pollutants.
The Future of Chemical Catalysts
Professor Cho’s assertion that this represents the first interaction of a manganese(III) complex with two coordinated water molecules reacting with aromatic hydrocarbons at lower temperatures speaks volumes about the potential for industrial applications. By fine-tuning the reduction potential of manganese, the researchers are inviting a new era of metal catalysts capable of breaking down even the most robust carbon-hydrogen bonds under mild conditions. Not only does this innovation provide a blueprint for sustainability, but it also serves as a reminder of the incredible possibilities that arise when we look to nature for inspiration in solving modern problems. As this research gains traction, its relevance in creating not only environmentally friendly technologies but also innovative industrial practices cannot be overstated. The future of pollution reduction may very well lie within this exciting discovery, and the prospects are nothing short of optimistic.