In the world of organic chemistry, the precision with which molecules can be manipulated is akin to an artist carefully balancing colors on a palette. The recent breakthrough by Professor Max Martin Hansmann and his research team signifies a monumental leap in this artistic endeavor, offering an innovative reagent capable of selectively introducing carbon atoms into molecular structures. Published in the prestigious journal *Science*, this study pushes the boundaries of traditional organic synthesis, heralding diverse possibilities for drug development and complex molecule formation.
The Genius Behind the Discovery
At the heart of this groundbreaking research is Professor Hansmann’s 2022 European Research Council (ERC) Starting Grant, which has propelled his work in the chemical sciences. Collaborating closely with Dr. Taichi Koike, a gifted postdoctoral researcher and Alexander von Humboldt Fellow, the research exudes confidence and foresight. As Hansmann aptly states, “The precise modification of molecules at a single-atom level is one of the most elegant transformations in organic chemistry.” The effectiveness of this approach hinges not just on theoretical prowess, but on practical applications that address real challenges facing chemists today.
A New Era for Carbon Atom Sources
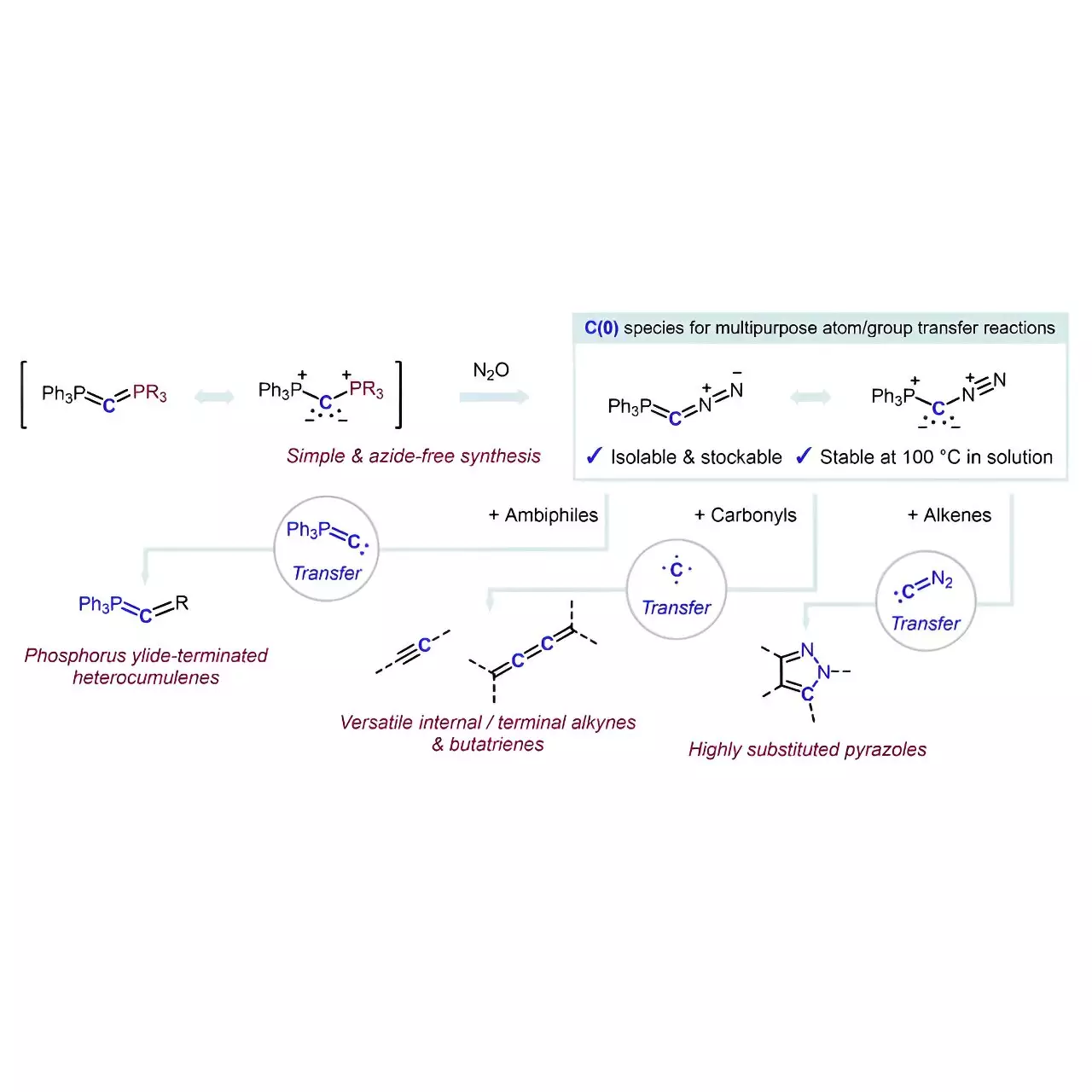
The introduction of this novel reagent, Ph3P=C=N2, is notable, given the complexities historically associated with selectively transferring carbon atoms in organic molecules. Researchers have long grappled with the difficulties of developing such reagents, often compounded by the inherent risks of traditional methods involving diazo compounds and azides. However, the Hansmann team’s process employs an elegant strategy that stands apart—it utilizes the simple coordination of a carbon atom with labile groups like PPh3 and N2, circumventing the safety issues linked with previous practices.
Exploring the Mechanism of Action
The innovation doesn’t merely end with the synthesis of Ph3P=C=N2. The research meticulously outlines how the reagent functions as a highly selective catalyst that facilitates a range of carbon atom transfers without necessitating auxiliary additives. What sets this reagent apart is its multifaceted utility: it serves as both a carbon atom source and a reagent for complex reactions, leading to the formation of invaluable chemical structures. The capacity of this regent to convert ambiphiles into phosphorus ylide-terminated heterocumulenes or to transfer N2 to alkenes highlights its versatility.
Potential Transformations in Pharmaceuticals
The implications of such a reagent extend far beyond academic curiosity; they potentially revolutionize the route to synthesizing intricate pharmaceuticals. Traditional synthetic sequences can be time-consuming and resource-intensive. However, the implementation of this new reagent could streamline the synthetic process, making it considerably shorter and more efficient. By allowing researchers to access complex molecules more straightforwardly, the pathway to innovative drug development is effectively paved.
A Bold New Direction for Research
What makes this discovery even more compelling is the commitment to its exploration. Professor Hansmann expresses a forward-looking confidence that further investigating the reactivity of this carbon transfer reagent will unveil novel applications and functionalities. The potential for accessing higher cumulenes and enhancing late-stage functionalizations of complex molecular architectures sparks excitement not only among chemists but in the broader scientific community as well. It’s a testament to the ingenuity required to confront and overcome the longstanding challenges in organic synthesis.
In sum, the research accomplished by Professor Hansmann and his team represents a significant achievement in the field of organic chemistry, characterized by both its innovative methodology and its transformative potential. As the scientific community begins to explore the myriad possibilities presented by this reagent, the future of organic synthesis appears brighter, promising advancements that could redefine the landscape of chemical research and development. The breakthrough stands as a vivid reminder of why curiosity, creativity, and scientific rigor must exist in tandem to drive progress and innovation in the dark realms of molecular complexity.