In an era wrestling with growing carbon footprints and climate change, the quest for sustainable energy solutions has never been more critical. For decades, chemists have been striving to convert waste materials into valuable resources. However, recent research signifies a paradigm shift by introducing the innovative use of electricity to refine these processes. A groundbreaking study published in Nature Catalysis highlights a remarkable achievement: the conversion of carbon dioxide, one of the planet’s most notorious greenhouse gases, into methanol, a potent liquid fuel. This development is not merely a scientific milestone; it has the potential to change the course of renewable energy.
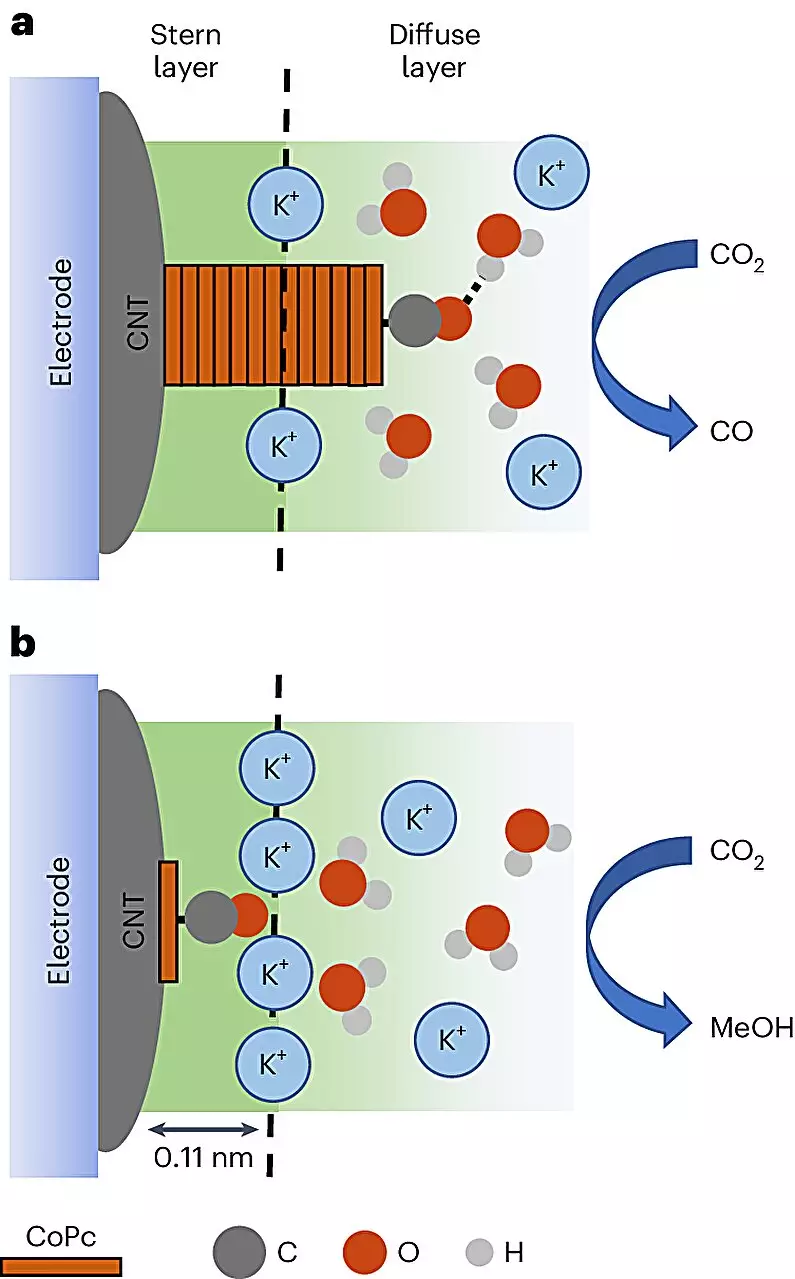
The researchers conducted this transformative process using cobalt phthalocyanine (CoPc) molecules, which were strategically applied to a surface of carbon nanotubes. These nanotubes possess exceptional electrical characteristics, making them the perfect medium for conducting electricity in a chemical reaction. By applying an electrical current to an electrolyte solution encompassing these nanoparticles, the team demonstrated a mechanism that allows CoPc to extract electrons and initiate the conversion of carbon dioxide to methanol. This clever interplay of electricity and chemistry could be the ticket to making energy production more sustainable and efficient.
Shedding Light on Catalytic Processes
One of the most exciting aspects of this study is its use of advanced in-situ spectroscopy techniques, providing unprecedented visual insight into the chemical reactions at play. For the first time, scientists were able to directly observe how CoPc molecules can either transform carbon dioxide into methanol or, regrettably, into carbon monoxide—an undesired byproduct. The environment surrounding each reaction significantly influenced these pathways. By adeptly managing the distribution of the CoPc catalyst across the carbon nanotubes, researchers found they could substantially amplify the likelihood of methanol production, increasing the efficiency eightfold.
This nuanced understanding opens new avenues for the field of catalysis. Previously, the optimization of such materials relied on empirical data without truly grasping the molecular mechanisms involved. The research signals a move toward integrating thorough scientific comprehension of these complex chemical interactions, paving the way for more effective catalysts and improved energy solutions moving forward.
The Vital Role of Interactive Chemistry
Diving deeper, the study reveals an intriguing interaction between CoPc molecules and highly charged particles known as cations, which appeared to enhance methanol formation. This discovery raises several compelling questions about the role these cations may play in the catalytic process overall. Given the importance of these interactions in maximizing efficiency, further research is essential to unlock their full potential. Such breakthroughs could establish a refined approach to not just methanol production but potentially a range of renewable fuels derived from waste materials.
Robert Baker, a co-author and chemistry professor at The Ohio State University, aptly reflects on the significance of this research: “We are witnessing systems of great importance and gaining insights into long-standing scientific uncertainties.” Recognizing the complexity of molecular reactions is imperative to effectively advance applications in renewable energy.
Methanol: A Game Changer for Renewable Energy
Methanol itself is among the most desirable products produced through this innovative catalytic process, primarily due to its high energy density. It could serve as a direct alternative fuel for various transport modes, from cars and airplanes to shipping vessels. Moreover, methanol generated from renewable electricity has the potential to power heating systems, electrical grids, and even fuel future chemical innovations.
The implications of this research stretch far beyond immediate fuel applications. It signifies a transformative step toward sustainable practices in energy production. If further investigation corroborates these findings and enhances our understanding of catalytic efficiencies, we could witness a robust shift in how we approach waste management and energy sustainability.
In a time where our dependence on fossil fuels is being scrutinized, the implications of this electric catalysis could not be more timely. As researchers continue to unearth the intricacies of molecular behavior in catalysts, we are on the brink of unlocking a treasure trove of potential solutions for some of our most pressing environmental challenges. The future appears vibrant with promise, not just for chemists or scientists but for everyone invested in a sustainable world.