The invention and mass production of plastics since the mid-20th century have led to an unprecedented global challenge. With around 8.3 billion metric tons of polymers generated, our planet is grappling with the consequences of this staggering output. Unfortunately, only a fraction—600 million metric tons—has found its way into the recycling stream. Instead, countless polymers have been relegated to landfills or incinerated, severely polluting our environments. As these materials linger in ecosystems for generations, it becomes increasingly clear that traditional recycling methods are failing us, and new strategies are desperately needed.
Groundbreaking Research from The University of Akron
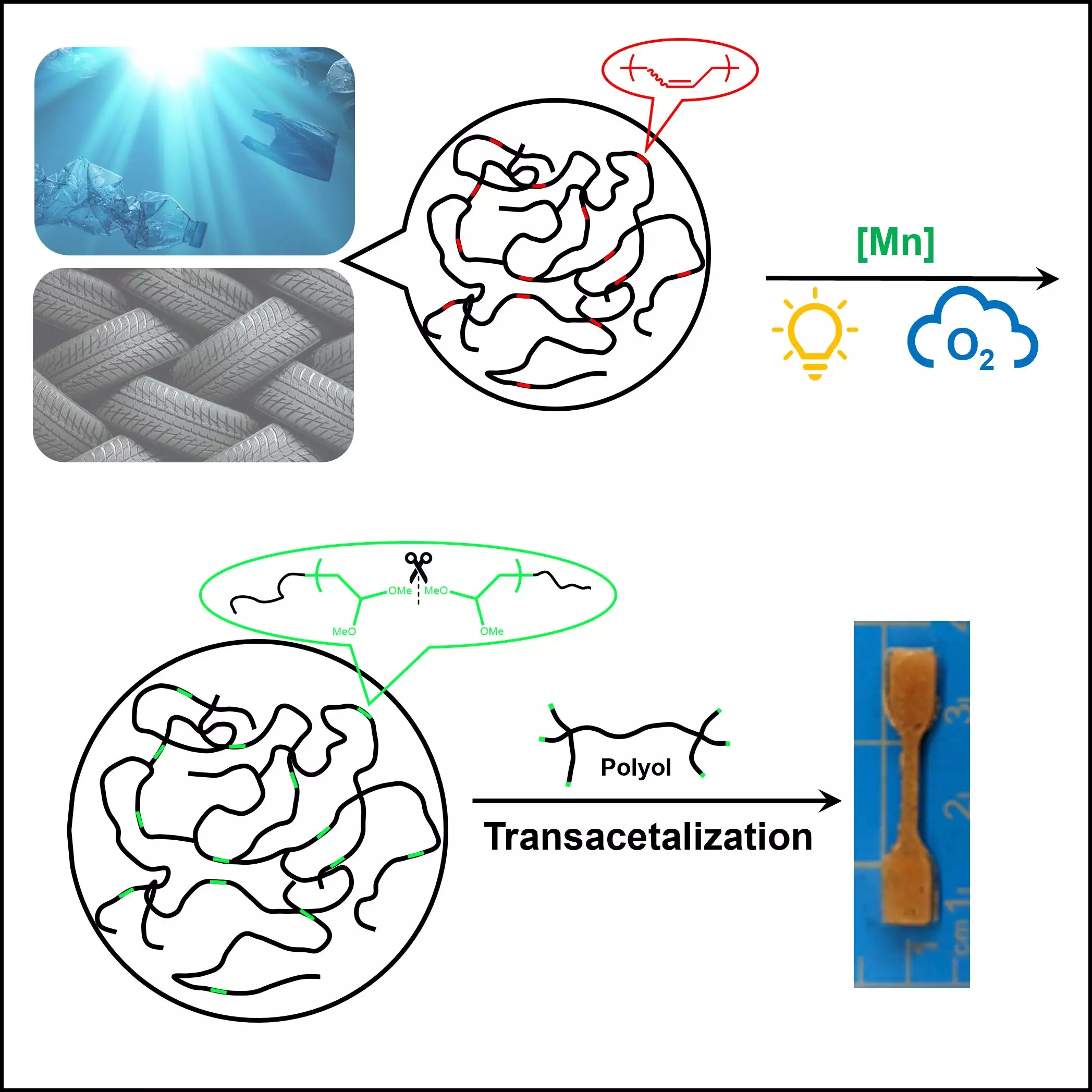
In the midst of this environmental turmoil, a recent breakthrough from Dr. Junpeng Wang and his team at The University of Akron provides a glimmer of hope. Their research, published in *Cell Reports Physical Science*, unveils an innovative approach to recycle unsaturated polymers, such as rubber and plastics, by harnessing the power of light and oxygen. Unlike previous methods, this process emphasizes a more natural breakdown of materials that contribute significantly to global plastic waste.
Wang’s work takes a critical look at the stability of polyolefins, which are the backbone of over 50% of all plastic production. The challenge posed by the resilient hydrocarbon structures inherent in these polymers has made recycling them incredibly difficult. Prior methods—such as ozonolysis and permanganate oxidation—while effective, have proven to be energy-intensive and environmentally detrimental. This insight into polymer composition led Wang’s team to explore unsaturation as a means to boost reactivity, presenting recycling opportunities that had previously been ignored.
A Catalyst for Change: Innovative Techniques
What sets this research apart is the introduction of a catalyst that, when activated by light, enables polymer breakdown at room temperature without the need for extreme conditions. This advancement marks a significant step forward, presenting a more sustainable method that aligns with our environmental goals. Utilizing abundant and green oxygen gas rather than harsh chemicals not only makes this process safer but also reduces ecological impacts often associated with traditional polymer degradation methods.
Unlike earlier strategies that produced hazardous by-products, Dr. Wang and his colleagues have created a controlled reaction that holds promise for scalability. This work pushes boundaries and provides an exciting opportunity for industries that have long struggled with polymer waste management.
The Road Ahead: Towards Sustainable Solutions
The implications of this research are vast, opening a path towards more responsible waste disposal and material recovery in the polymer sector. As the world faces increasing environmental challenges, it is paramount that we support innovations like this that propel us toward sustainability. Wang’s team is not merely tackling polymer waste; they are rethinking the future of plastics, making what once seemed like an insurmountable obstacle into a manageable opportunity.
The journey to a more sustainable world may be fraught with complexity, but with dedicated research and innovation, we inch closer to solutions that could transform how we interact with materials that have defined modern life. When scientists like Dr. Wang prioritize environmentally sound methods, they not only address current issues but inspire a new era of conscious consumerism and manufacturing practices.